About Us
Welcome to Brain Health Company
BRI Pharma contributes to solve unmet medical needs especially for the brain health field including AD, DLB and related brain inflammatory diseases.
Who Are We
BRI Pharma evaluates risk of dementia with blood and supplies the preventive tools of dementia.
Our Mission
The drugs prevent accumulation of misfolding proteins accounting Alzheimer’s, Parkinson’s and Lewy body diseases
What We Do
R&D for brain drugs
Evaluation of drug candidates with novel animal models
Licensing & Marketing
Providing diagnosis technologies
Tech Support
Company Profile
Company
HQ Address
Kagitori 3-2-20-104, Taihaku, Sendai, Miyagi, 982-0804 Japan
Lab. Address
Department of CNS drug innovation, Tohoku University School of Pharmacy, Aramaki Aoba 6-3, Aoba Sendai 980-8678, Japan
Branch Address
Mitsugi 53, Hayato Kirishima 899-5117, Japan
President&CEO
Kohji Fukunaga
# of Director
2 ppl.
Business Field
R&D and licensing of pharmaceutical product candidates and diagnosis technologies.
Date of Establishment
May 13, 2021
TEL&FAX
TEL: 022-796-2508, FAX:022-796-2508
Management Team
Kohji Fukunaga Ph.D., CEO
After graduating from the Graduate School of Pharmaceutical Sciences, Kumamoto University in 1980, Dr. Kohji Fukunaga obtained a doctor of medicine. In 1982, he discovered CaM kinase II (CaMKII), the role of CaMKII in spine dysplasia of mental retardation, regulation of dopamine D2 receptor function by fatty acid-binding protein (FABP), and Lewy’s body disease by FABP. In addition to his neuroscience, he has also contributed to drug discovery science, including the development of seeds for the treatment of Alzheimer’s disease and Lewy body disease targeting CaMKII and FABP. In his academic activities, he served as the editorial chairman and director of the English journal, Journal of Pharmacological Sciences, for a long time, and made a great contribution to the Japanese Pharmacological Society. His enormous research results so far have been commercialized and he is now active in the drug discovery business.
Tetsuro Nishi, CFO
After graduating from School of Economy in Kokugakuinn University in 1978. He started work in the bank of Kagoshima Shinkin. In 2010, he had appointed council member of Kagoshima Shinkin by 2019. He has licenses of financial planner and DC planner.
Scientific Advisors
Ichiro Kawahata Ph.D.
Graduate school of Pharmaceutical Sciences, Tohoku University
He obtained a doctorate (science) from the Graduate School of Life Science and Engineering, Tokyo Institute of Technology in 2009. He is working on the etiology of Parkinson’s disease and development of therapeutic agents by the discovery of novel molecular mechanism that controls dopamine function, and exploration research of new drug discovery targets. In addition, he is also engaged in drug development research for dementia including Alzheimer’s disease and Lewy body dementias.
Professor David I Finkelstein, Ph.D.
Head, Parkinson’s Disease Laboratory, Florey Institute of Neuroscience and Mental Health
Prof. David Finkelstein trained as a neurophysiologist, giving him a broad systems wide approach to health and disease. David is on editorial boards for a number of scientific journals and has published over 200 articles on the biology of Parkinsons and Parkinsonism. Over the last 5 year two compounds developed in conjunction with Biotech, PBT434 (Alterity Biotechnology) and Cu(ATSM) (Collaborative Medicinal Development), have safely been tested in people (phase 1 trials) and further trials are planned in the near future. David is on the Board of Parkinson’s Victoria and organisation dedicated to improving the lives of people living with Parkinson’s.
Professor Motohiro Morioka, M.D., Ph.D.
Neurosurgery of Kurume University
He obtained a medical doctorate from the Graduate Kumamoto University School of medicine in 1988. He was appointed associate professor of Department of Neurosurgery, Kumamoto University School of Medicine in 2005, and appointed to a professor of Department of Neurosurgery, Kurume University School of Medicine in 2011. He is familiar with all fields of neurosurgery and supplies the surgical techniques. Furthermore, he has been working on research of cerebral blood flow and metabolism and on therapeutic drug development for cerebral ischemia and related diseases.
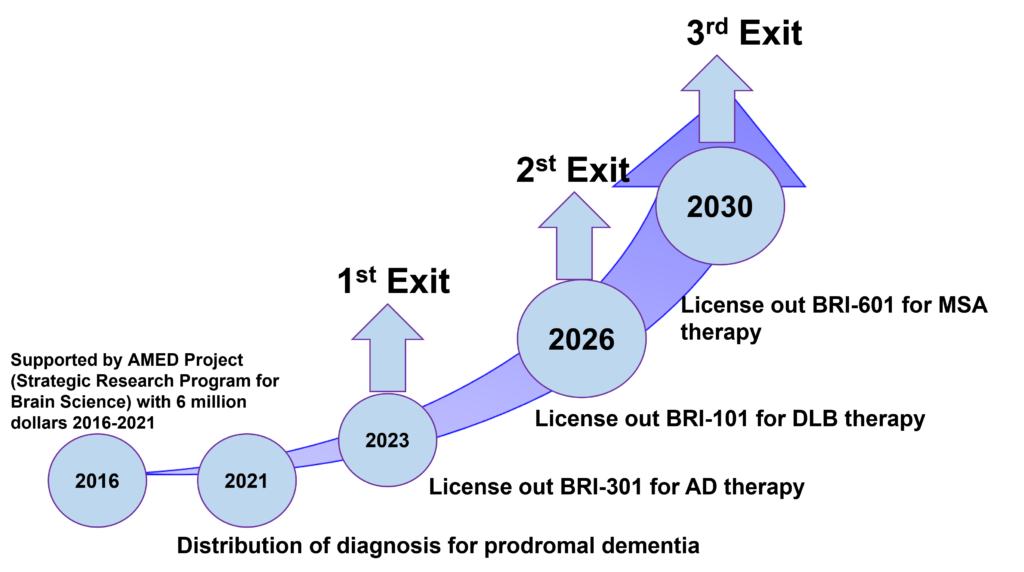
Our Business
We are promoting the commercialization of university-originated technologies. We have technological seeds in the field of brain diseases and we will proceed with the initial clinical development of drug candidates and aim to out-license them. On the other hand, we are also developing dementia diagnosis technology and will promote the commercialization of diagnosis services.